sandwich; 7 strands in 2 sheets, greek-key
variations: some members have additional 1-2 strands
contains copper-binding site
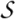 uperfamily
uperfamily
consists of three domains of this fold
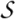 uperfamily
uperfamily
|
|
Generated from scop database 1.75 with scopm 1.101 on Wed Jun 3 10:42:06 2009
Copyright © 1994-2009 The scop authors / scop@mrc-lmb.cam.ac.uk |